Retatrutide Dosage Guide
Key Points
- Retatrutide is not yet approved and is currently being studied in large Phase 3 clinical trials.
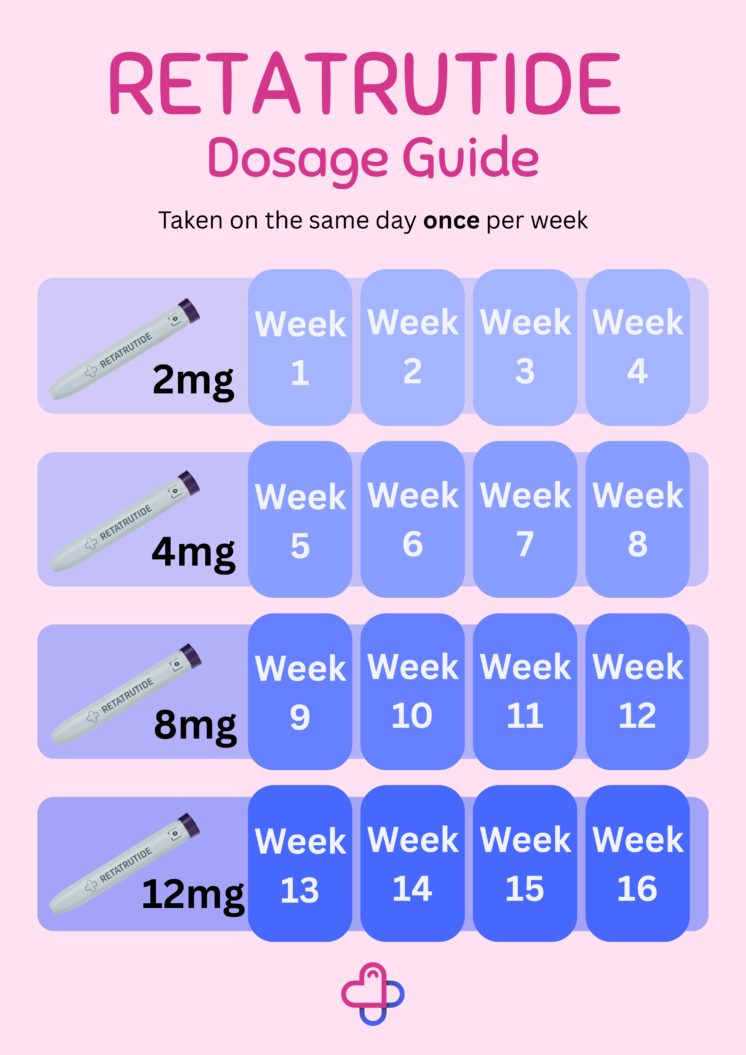
- Clinical studies have evaluated weekly doses of 1mg, 2mg, 4mg, 8mg, and 12mg.
- Most participants started at 2mg once weekly, with the dose gradually increased every 4 weeks to improve tolerability and effectiveness.
- The 12mg dose produced the greatest weight loss, with participants losing up to 28.7% of their body weight after 68 weeks.

Written By Chemist Click
Reviewed By Pharmacist Abbas Kanani
Last updated on 7th April 2026 • 5 minute read