Retatrutide has produced some of the strongest weight loss results seen in obesity drug trials so far.
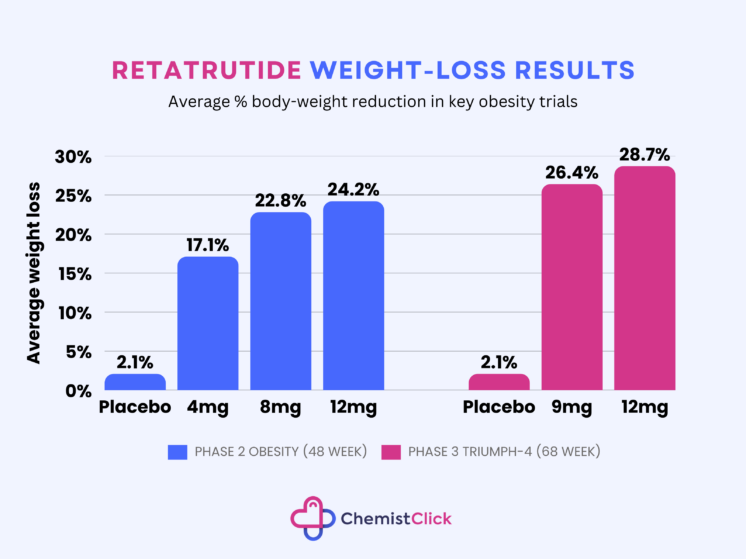
In the Phase 3 trials, adults taking retatrutide 12mg lost an average of 28.7% of their body weight after 68 weeks.
Weeks 1-4: Month One
In the first month, weight loss is likely to be gradual. Retatrutide is started at a lower dose so the body can adjust and side effects can be managed.
You are likely to notice:
- Reduced hunger
- Feeling full sooner
- Smaller portion sizes
- Fewer cravings
- Early weight loss
You can expect to lose around 2-5% of your starting weight after one month on retatrutide.
However, major changes on the scales are more likely to build after several weeks or months.
Weeks 5-8: Month Two
By month two, appetite suppression may become more noticeable, especially as the dose is increased. This is when some people may start to see more consistent weight loss.
You may find it easier to eat smaller meals, avoid snacking and stay in a calorie deficit. Side effects such as nausea, bloating, constipation or diarrhoea may also be more noticeable during dose increases.
After two months on retatrutide, you can expect to lose around 4-8% of your starting weight.
Weeks 9-12: Month Three
By month three, weight loss may become more visible. Clothes may feel looser, waist measurements may reduce, and appetite control may feel stronger.
This is also when lifestyle habits become more important. Retatrutide may reduce hunger, but the best results are likely to come when it is combined with a balanced diet, regular movement and enough protein.
After three months on retatrutide, you can expect to lose around 6-10% of your starting weight.
Weeks 13-24: Month Six
By six months, retatrutide results may be significant for people who respond well to treatment.
In earlier Phase 2 data, people taking retatrutide for six months lost up to 17.5% of their body weight at 24 weeks.
Weeks 25-52: One Year
By one year, results may be substantial.
Phase 2 trial data showed average weight loss of up to 24.2% at 48 weeks, while the newer Phase 3 trial data showed 28.7% average weight loss at 68 weeks on the 12mg dose.
This suggests that weight loss may continue to build beyond 12 months for some people.