Orforglipron vs Wegovy Tablets
Key Points
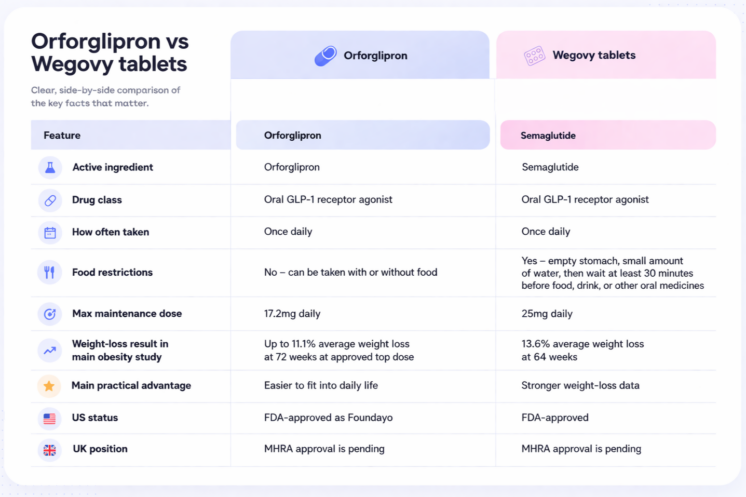
- Results: In trials, Wegovy tablets have shown greater average weight loss (~13%) compared to orforglipron (~11%).
- Administration: Orforglipron does not have food or water restrictions. Wegovy tablets need to be taken in the morning on an empty stomach with water.
- Side effects: Both medicines mainly cause gastrointestinal side effects such as nausea, diarrhoea and constipation.

Written By Chemist Click
Reviewed By Pharmacist Abbas Kanani
Last updated on 14th May 2026 • 5 minute read